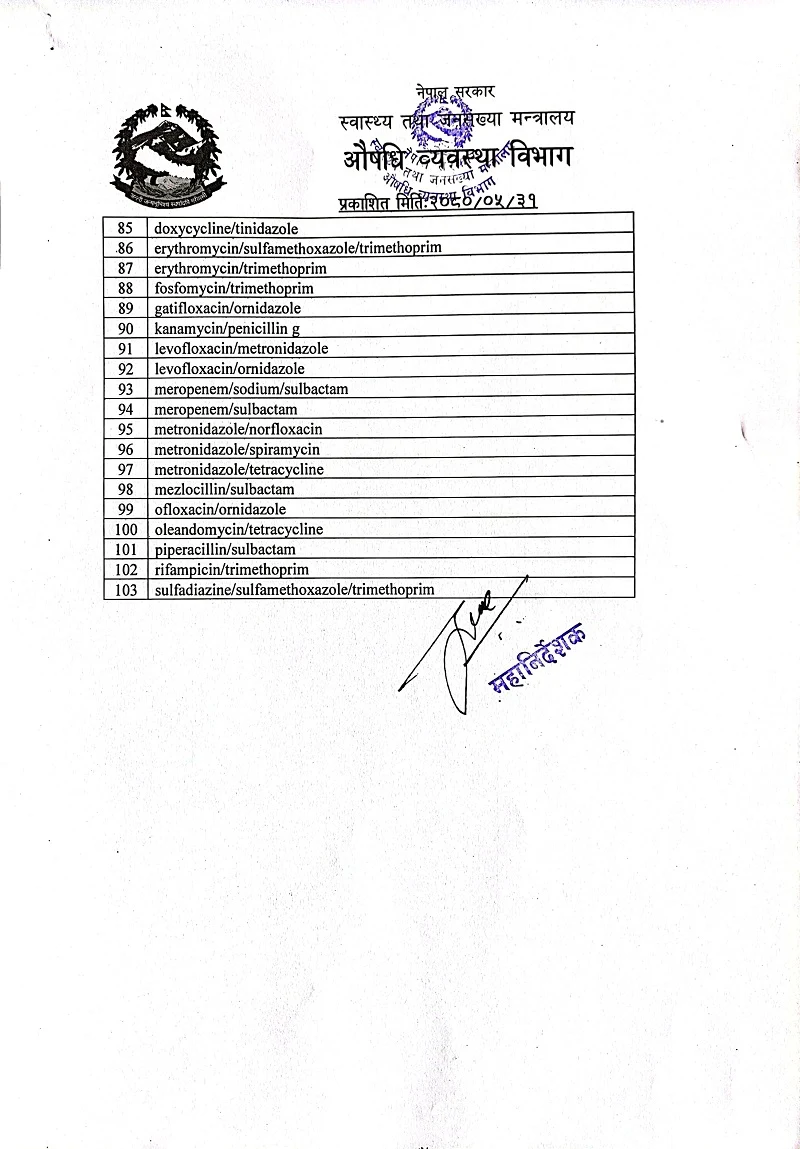
The government of Nepal has instructed consumers and stakeholders not to use one hundred and three types of antibiotics.
Narayan Prasad Dhakal, director general of the Department of Drug Administration (DDA), said that he has given instructions to minimize the use of antibiotics and not to use them for prevention and control. Even the World Health Organization (WHO) has not recommended the use of those medicines. He also said that there is no reason to use those antibiotics.
"WHO has published a list of 103 types of antibiotics that should not be used", he said, "we neither register those drugs nor give permission for use." According to him, 13 types of antibiotics have been registered out of 103.
The department has said that a national action plan has been prepared and approved to effectively reduce the morbidity rate, death rate, and economic impact caused by antibiotics in accordance with a health concept in accordance with the global action plan of such antibiotics.
Director General Dhakal said that in order to achieve the objective of promoting an evidence-based treatment system and proper use of antibiotics, the Drug Advisory Committee has instructed to register, renew, import, and not/not use those drugs.
Combinations of antibiotics that are registered in the department and not recommended by the WHO and new registration of raw materials for the purpose of producing the combination of those antibiotics, additional registration of new entry and no entry recommendation certificate, production license of the combination of such antibiotics registered in the department, market sale distribution certificate It has been decided not to renew the adoption registration certificate, adoption recommendation letter.
Raw materials and primary and secondary packing materials of such types of antibiotics registered in the department are requested to be submitted to the inventory department and effective from the date of the decision, the domestic industry will not produce more of such types of drugs, and importers are also requested not to import more antibiotics.
Dhakal, the director general of the department, informed that when there are 13 types of antibiotics registered in the department, they will be allowed to sell and distribute them only for six months.
According to him, it has been decided to make arrangements so that the non-recommended antibiotics and combinations that are currently produced or imported and available in the market or are in the production stage will be available in the market for a maximum of six months from the date of the decision, i.e. on the 4th of September.